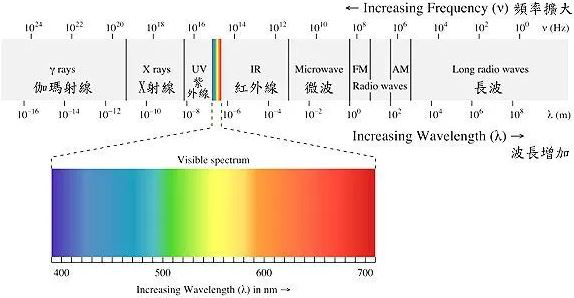
Visible Spectrum (可见光谱)
The visible spectrum is the range of light that human vision can perceive. When white light passes through a prism or diffraction grating, it produces a continuous spectrum of colors, including red, orange, yellow, green, blue, indigo, and violet. In the visible region, there are also line spectra and band spectra. It's a very small region within the entire electromagnetic spectrum.
The entire electromagnetic spectrum includes radio waves, infrared, ultraviolet, and X-rays, among others. These waves have different wavelengths, with wavelengths between 400 and 760 nanometers being typical for visible light.
Visible Light Sources
The primary natural source of visible light is the Sun, while the main artificial sources are incandescent objects (especially incandescent lamps). The visible light spectrum emitted by these sources is continuous. Gas discharge tubes also emit visible light, with their spectra being discrete. Various gas discharge tubes are often used in combination with filters as monochromatic light sources.
The range of light visible to the human eye is influenced by the Earth's atmosphere. For most electromagnetic radiation, the atmosphere is opaque, except for the visible light spectrum and a few exceptions like radio communication bands. Many other organisms can see light waves differently from humans; for example, some insects, including bees, can see ultraviolet light, which is helpful in finding nectar.
The spectrum does not include all colors that the human eye and brain can distinguish, such as brown, pink, and magenta, as they require a mixture of multiple wavelengths to adjust the intensity of red.
The wavelength of visible light can penetrate optical windows, meaning it can pass through the Earth's atmosphere with minimal attenuation (blue light scattering is more severe than red light, which is why we see the sky as blue). The human eye's response to visible light is subjectively defined (see CIE), but the atmospheric window is defined using physical measurements. It's called the visible light window because it covers the spectrum visible to the human eye. The near-infrared (NIR) window lies just outside the human eye's visible range, while the mid-wave infrared (MWIR) and far-infrared (LWIR, FIR) windows are further away.

Applications
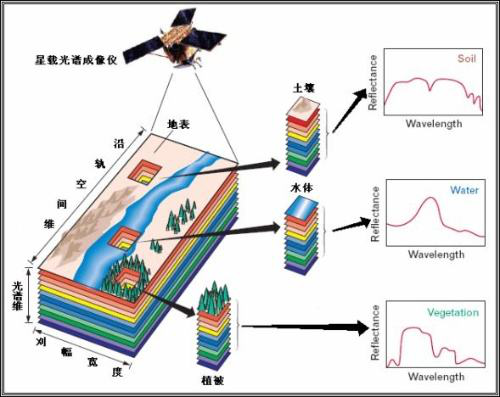
Visible spectrum imaging systems include light source systems, spectroscopic systems, image imaging and recording systems, image analysis and processing systems, etc. They consist of optical lenses, liquid crystal tunable filters (LCTF), CCD cameras, light sources, and computers, among other devices. The core component is the liquid crystal tunable filter, which functions similarly to a high-quality band-pass interference filter. With the adoption of electronic control liquid crystal adjustment methods, problems such as uniformity within the light transmission surface, peak spectral transmittance, and out-of-band suppression have been solved. This demonstrates high precision and easy real-time control, playing a positive role in the application of spectral imaging technology.
Commercial instruments for visible spectrum imaging systems mainly include the Nuance multispectral imaging system from CRI in the United States and the Condor hyperspectral imaging system from ChemImage in the United States. The spectral working range of this system is 410 nm to 720 nm, with a spectral resolution of less than 10 nm, an imaging field of view of approximately 260 mm x 250 mm, and an enlarged field of view through optical components. In addition, researchers such as Xu Xiaoxuan and Shen Zhixue in China have designed and developed visible light liquid crystal spectral imaging systems with simple structures, high spatial resolution, and high spectral resolution.
Infrared Spectrum (红外光谱)
Infrared spectrum refers to the selective absorption of certain wavelengths of infrared radiation by molecules, causing transitions in vibrational and rotational energy levels within molecules. The detection of infrared absorption can yield the infrared absorption spectrum of a substance, also known as a molecular vibration spectrum or vibration-rotation spectrum.

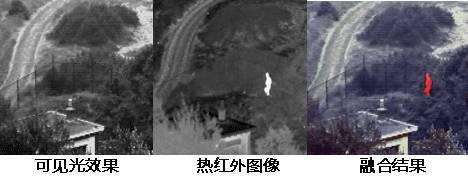
Zones of Infrared Spectrum
The infrared spectrum is typically divided into three regions: near-infrared (0.75-2.5 μm), mid-infrared (2.5-25 μm), and far-infrared (25-300 μm). Generally, the mid-infrared region, where most organic and inorganic compounds' fundamental absorption bands occur, is the most studied and applied region, with the most accumulated data and mature instrument technology. The commonly referred to infrared spectrum usually means the mid-infrared spectrum.
Applications
The applicability of infrared spectrum to samples is quite extensive, including solid, liquid, or gaseous samples, and it can detect inorganic, organic, and polymeric compounds. Additionally, the infrared spectrum has characteristics such as rapid testing, convenient operation, good repeatability, high sensitivity, low sample consumption, and simple instrument structure. Therefore, it has become an essential tool in modern structural and analytical chemistry. Infrared spectroscopy has extensive applications in the study of the configuration, conformation, and mechanical properties of polymers, as well as in physics, astronomy, meteorology, remote sensing, biology, medicine, and other fields.
The positions and intensities of infrared absorption peaks reflect the characteristics of molecular structures and can be used to identify the structural composition of unknown substances or determine their chemical groups. The absorption intensity of absorption bands is related to the content of chemical groups and can be used for quantitative analysis and purity identification. In addition, infrared spectroscopy plays a certain role in the study of chemical reaction mechanisms. However, its most common application is the structural identification of unknown compounds.
Infrared spectroscopy has high specificity, so it is common to use a comparative method with the infrared spectra of standard compounds for analysis and identification. Several collections of standard infrared spectra have been published, such as the "Sadtler Standard Infrared Grating Spectrum Collection," which collects infrared spectra of more than 100,000 compounds. In recent years, some of these spectra have been stored in computers for comparison and retrieval.
Ultraviolet and Visible Spectrum (紫外和可见光谱)
Basic Principles
Generation of Spectra
In the ultraviolet spectrum, wavelengths are measured in nanometers (nm). The ultraviolet spectrum ranges from 10 to 380 nm and is divided into two segments. The range of wavelengths from 10 to 200 nm is called the far-ultraviolet region. This wavelength can be absorbed by nitrogen, oxygen, carbon dioxide, and water in the air, so it can only be studied in a vacuum. Thus, the absorption spectrum in this region is called vacuum ultraviolet, but due to high technical requirements, its use in organic chemistry is limited. The range of wavelengths from 200 to 380 nm is called the near-ultraviolet region, and the absorption spectrum in this region is generally referred to as ultraviolet spectrum. The range of wavelengths from 400 to 750 nm is called the visible spectrum. Most spectrophotometers include both UV and visible parts, typically covering a range of 200 to 800 nm (or 200 to 1000 nm).
Spectral Lines
Many organic molecules undergo electronic transitions, absorbing light in the range of 200 to 1000 nm, which falls within the ultraviolet-visible light region. Therefore, ultraviolet absorption spectra are generated due to transitions of valence electrons in molecules, also known as electronic spectra.
Applications
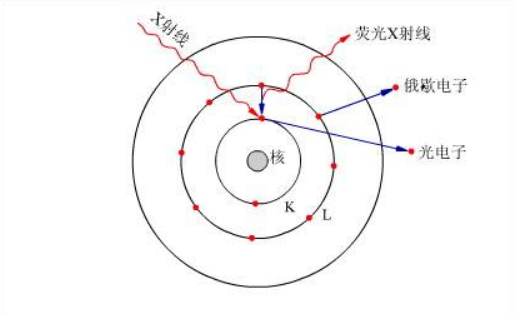


In medicine, ultraviolet spectra have played an important role in the breakdown of a series of vitamins, antibiotics, and natural products' chemical structures, such as vitamins A1, A2, B12, B1, penicillin, streptomycin, terramycin, and luminescent substances from fireflies.
Performance Testing: Photochromism refers to the reversible change in color under light irradiation. It can be tested and studied using ultraviolet spectroscopy.
Photochromic materials, as a type of new functional materials, have broad application prospects. For example, they can be used as light information storage materials, light switches, light converters, etc., with applications in mechanical, electronic, textile, defense, and other fields. The development of photochromic coatings, photochromic glass, and photochromic inks has practical application significance. In addition to the above applications, photochromic materials can also be used as self-developing photosensitive films, holographic photographic materials, protective and decorative materials, printing plates, printing circuits, camouflage materials, etc.
It is particularly noteworthy that the study of photochromic compounds as rewritable optical storage materials has become one of the hotspots in the field of photochromism in recent years. As a photochromic optical storage medium, it needs to meet certain conditions, such as absorption in the semiconductor laser wavelength range, non-destructive readout, good thermal stability, excellent fatigue resistance, and fast response speed.
FALenses Technology specializes in providing machine vision core hardware. You can go to the official website of FALenses Technology at https://www.falenses.com/ for more information.
